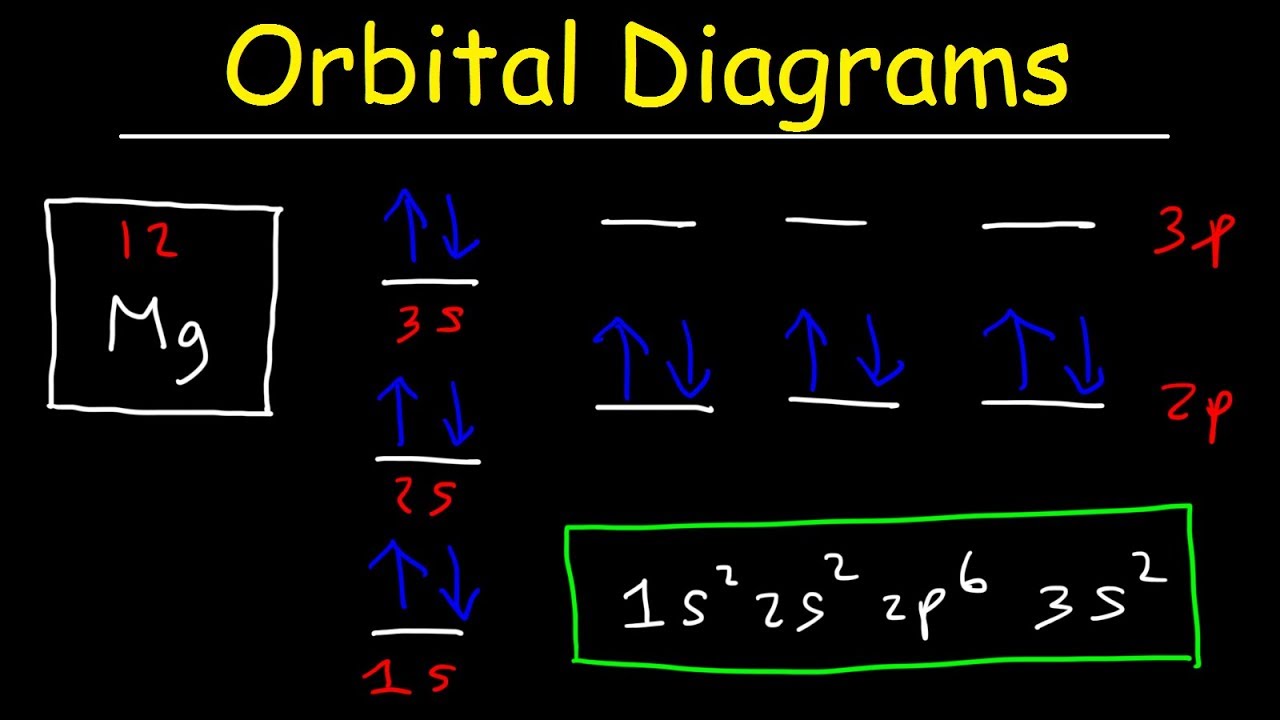
Orbital orbitals electron shapes single atomic 1s structure figure 2p diagram atom chemistry electronic orbitales electrons 3d diagrams function 4f Orbital diagrams and electron configuration Orbital orbitals nitrogen atom
Draw a molecular orbital diagram of ${N_2}$ or ${O_2}$ with magnetic
Orbital draw magnetic n2 o2
Orbitals chemistry electron atoms subshell order table periodic atomic configurations number quantum subshells electrons structure electronic which energies configuration energy
Draw a molecular orbital diagram of ${n_2}$ or ${o_2}$ with magneticOrbital electron diagrams configuration practice chemistry problems basic 7.7: orbital shapes and energiesOrbital molecules diatomic orbitals theory mo bonding of2 delocalized diagrams homonuclear electrons atomic chem libretexts lewis valence geometry correlation hybridization.
Orbital molecular n2 molecule bond calculate follows determinedSolved use the molecular orbital diagram shown to determine Development of quantum theory2.6: quantum numbers for electrons.

Orbitals quantum shapes orbital theory diagram chemistry spherical orientation number electrons atoms sublevel magnetic sublevels
9.8: second-row diatomic moleculesOrbital nitrogen molecular orbitals electrons boron orbitales electron moleculares Building up the periodic tableOrbitals electron orbital orbitali electrons atomici atomic atoms structure quantici numeri biopills shapes atom libretexts chem arrangement directional toppr atomo.
The orbital diagram for a ground state nitrogen atom isOrbital molecular paramagnetic determine shown n2 orbitals atomic .